Hydrocarbons - Alkanes
Page 1 of 1
 Hydrocarbons - Alkanes
Hydrocarbons - Alkanes
For an introduction on Hydrocarbons.
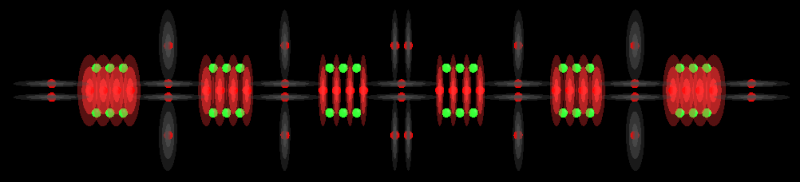
Alkanes have the general formula CnH2n+2.
You can find background information on Alkanes in this wiki page.
These models show the simple forms of the Alkanes. It is possible for 3 Carbon atoms to bond at the same location which create isomers. As the chain gets longer, the number of possible isomers increases dramatically but I will try to cover some of the basic ones later. I also won't show Methane because Miles has already done so and it is a special case that does not fit into this structure.
Alkanes have the general formula CnH2n+2.
You can find background information on Alkanes in this wiki page.
These models show the simple forms of the Alkanes. It is possible for 3 Carbon atoms to bond at the same location which create isomers. As the chain gets longer, the number of possible isomers increases dramatically but I will try to cover some of the basic ones later. I also won't show Methane because Miles has already done so and it is a special case that does not fit into this structure.
Last edited by Nevyn on Mon Nov 17, 2014 6:36 am; edited 1 time in total
 Similar topics
Similar topics» Hydrocarbons
» Hydrocarbons - Alkenes
» Hydrocarbons - Arenes
» Hydrocarbons - Alkynes
» Hydrocarbons - Cycloalkanes
» Hydrocarbons - Alkenes
» Hydrocarbons - Arenes
» Hydrocarbons - Alkynes
» Hydrocarbons - Cycloalkanes
Page 1 of 1
Permissions in this forum:
You cannot reply to topics in this forum|
|
|